NOT SURE IF YOUR PRODUCT QUALIFIES AS A FEED ADDITIVE OR WHICH CATEGORY TO APPLY FOR?
In the EU, feed additives are defined as products which are intentionally added to feed or water in order to favourably affect the quality of feed and the quality of food from animal origin.
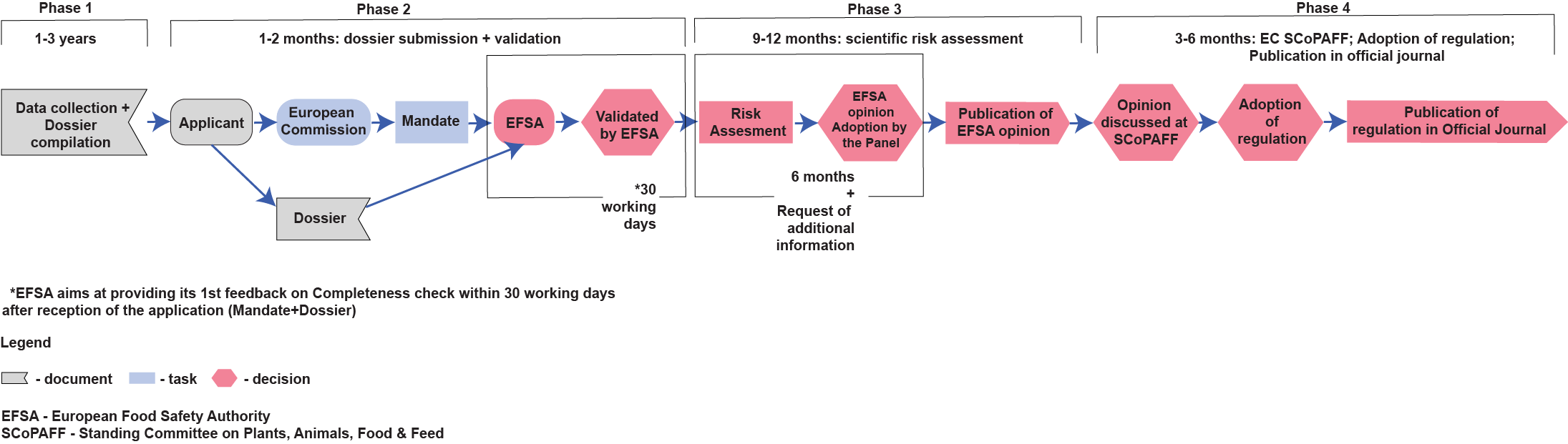
In order to protect human health, animal health and the environment, feed additives must undergo a pre-market safety assessment via a centralised procedure following Regulations (EC) Nº 1831/2003 and 429/2008 before being placed on the EU market. The scientific safety assessment is carried out by the European Food Safety Authority (EFSA) and the authorisation of the additive is granted by the European Commission (EC).
They typical feed additive evaluation process is outlined below:
For more information on UK authorisation, please visit this web page.